| To be presented at "Towards a science of consciousness"
conference April 2012 Tucson Arizona by
Rolf Ekedahl M.D., Ph.D. Clinical Neurophysiology NeuFyDi AB,
Stockholm, Sweden.
Introduction
Since anaesthesia and muscle blockade with assisted ventilation
was introduced during ECT it has become even more important to
monitor the cerebral electrical events.
The commonly used anaesthetics have a strong antiepileptic effect,
and therefore these anaesthetics will to some often unknown extent,
counteract the induced epileptically activity with the purpose of
triggering a generalized epileptically grand mal seizure.
The electrical stimulation devices commonly used Thymatron® (Somatics
Lcc) and MECTA® (MECTA corp.), have a simplified EEG-monitoring
system, which record and evaluate the EEG-activity from one channel
after the actual electrical stimulation. The filter settings (< 6Hz)
restricts it to monitor the low frequency response from the
epileptically seizure activity, namely the inhibitory activity. In
addition, the recording electrodes are located just above subdermal
muscles and this low frequency muscle activity can contribute to the
recorded signal.
Objective
We wanted to ensure recordings of the epileptically activity,
also the high-frequency components throughout the whole ECT-session
including the electrical stimulation sequence and to reduce
potential artefacts.
Method
The electrical stimulation at the ECT sessions were according to
standard procedure either with a bifrontal or RUL (right unilateral)
frontal stimulation (illustration 1), the stimulation device ,
Thymatron®, delivered square pulses with 0,25 -1 ms (4000-1000 Hz)
and with a stimulus pulse interval between 40-140 Hz.
In order to avoid the pitfalls described above with the
monitoring technique alternative recording positions were chosen
namely P3, P4 and the reference position CZ used in the 10-20 system
for EEG. These positions were chosen to avoid closeness to
underlying subdermal muscle activity and to ensure recordings during
the stimulation.
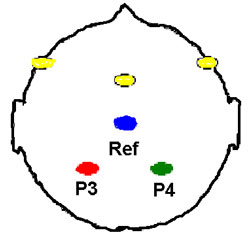
Click to enlarge illustration 1.
Illustration 1: The cortical regions, which were recorded and
stimulated seen from above with the nose pointing upward. The yellow
dots indicate the stimulation positions were either between the
laterally frontal positions (bilateral stimulation) or between the
midfrontal and right lateral frontal position (RUL- right
unilateral). The recordings were positioned at P3 and P4 with
reference position Cz (10-20 EEG-systems). In these study only
recordings from P4 with reference, Cz is presented.
The digital EEG-system Nervus 5.3 (Viasys Healthcare™) was used
for recording and analyzing the recorded EEG. The digital amplifier
had a sampling frequency of 128 Hz, the digital filters used were
set to 5 Hz low-cut and off fore high-cut, and a notch filter set at
50 Hz.
The frequency was analysed for the recorded signal throughout the
stimulus at the P4 site using Cz as reference. The obtained
frequency patterns were further analysed and divided in to two
frequency bands, low-high frequency between 5 and 30 Hz (α
and β EEG-frequencies) and high frequency
from 30 Hz (γ EEG-frequency) up
to 100 Hz. According to the percentage of gamma (γ-activity
(30-100 Hz, 50 notch filtered), the individual ECT-sessions were
plotted and correlated to the frequency pattern.
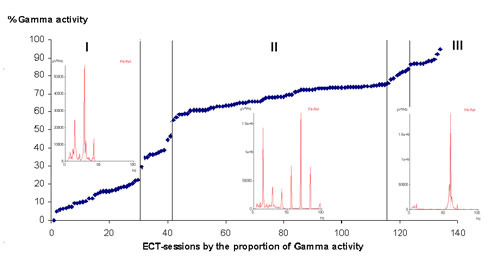
Click to enlarge illustration 2
Illustration 2: 134 individual ECT-sessions (x-axis) in the
diagram (dark blue markings). At the Y-axis is the percentage of
Gamma- activity (>30Hz <100Hz). Three groups of frequency responses
could be distinguished (I-III) which were correlated to the amount
of Gamma (γ) activity. The lowest percentage of gamma (γ) activity classified to group
I and to group III the highest. The frequency
distribution differed between the groups shown in the three inserted
frequency diagrams showing the different peak distributions in
respectively group. In group I an oligo-peak distribution with up to
22 % gamma (γ) activity proportion, in group
II a multi-peak
distribution with 56-76 % gamma (γ) activity proportion and in group
III a mono-peak distribution with 86-94 % gamma (γ) activity
proportion. Between group’s I-II and II-III transformation zones
were the frequency distribution is a mixture of described frequency
pattern in group I and II respectively II and
III.
Results
The analyzes of the 134 individual ECT-sessions showed a varying frequency pattern and also a much varying content of gamma (γ) activity varying from 5-94 % percentage proportion
(illustration 2). There were not a stereotyped generalized epileptically activity which could have been expected. According to the different frequency patterns and content of gamma activity the resulting responses were subdivided into three different groups here called
I-III.
The first group I showed an oligo-peak frequency distribution with well distinguished peaks at 13,7 and 28,3 Hz and a gamma (γ)-activity proportion of 0-22 %. 29/134 (22%) of the ECT-sessions were classified to this first group and the proportion of gamma-activity were 5-22 %.
The second group II showed a multi-peak frequency distribution with peaks at regular intervals between 13,7-95,5 Hz (illustration 2). 74/134 (55%) ECT-sessions were classified to group
II and the proportion of gamma-activity was 56-76 %.
The third group III showed a splintered mono-peak distribution, with a dominating frequency around 60 Hz. 10/134 (7%) of the ECT-session was classified to group III and the proportion of gamma-activity was between 86-94 %.
Separating group I-II and II-III were transformation zones
(illustration 2) were the frequency distribution showed intermediate frequency distributions patterns between group I and II respectively
II and III.
The amplitude for the peaks had high amplitudes compared to normal EEG-amplitudes (<100
μV) and the highest in group
III with approximately 1000
μV.
Discussion
Technical aspects:
The technical drawbacks that easily could be raised are if actual cerebral electrical activity had been recorded but noise and artefacts from the electrical device and the stimulus pulse. To prevent unwanted 50 Hz activity when analyzing the signals, a 50 Hz notch-filter was used and the influence of this 50 Hz activity could be neglected. An other potential source for artefacts was from the stimulus pulse. The stimulus-pulse could be set to 0,25-1 ms pulse length, expressed in frequencies between 1000-4000 Hz for the Thymatron stimulus device used in this study, which are well above the 128 Hz sampling frequency for the recording system and would not contribute to the recorded activity. To avoid unwanted muscle activity the recording position was positioned at the Parietal region, P3, P4 and Cz (10-20 EEG-system) There are no underlying subdermal muscles in that area.
Clinical aspekts:
What can be expected from the electrical stimulation during ECT is that a strong generalized epileptically activity should be triggered as a generalized epileptically seizure follows the stimulation. Instead a hyper- synchronized Alfa (a), Beta (b) and Gamma (g)-activity were triggered and in a majority of the responses gamma (g)-activity occurred to a varying degree. This finding raises questions about which role gamma (g)-activity have in the treatment and adverse effects occurring because of Electroconvulsive treatment. Another clinical interesting observation is the graded responses to stimulation seen during the stimulation and if that can be used as a method to predict if a stimulation have had a satisfying effect for the result of the treatment.
Neurophysiological aspects:
In a majority of the responses a high amplitude hyper-synchronized gamma (γ)-activity was triggered from the frontal stimulation position and recorded at a distant parietal position. Most probably, this is a transmitted activity via Cortico-Cortico connections, Cortico-Thalamic pathways or association pathways. No firm conclusions on this issue can be drawn from this study, but some observations shows different responses in the left parietal region compared to the right and a time lag between these responses (inter-hemispheric pathways?). As the stimulation does not reflect a normal brain activity the relation to normal neurophysiologic processes is uncertain e.g. the transient moving
gamma (γ)-activity suggested by Professor Stuart Hameroff as a basis for conscious cognitive neurocomputation (The “consciousness pilot”-dendritic synchrony moves through the brain to mediate consciousness. Stuart Hameroff., Journal of Biological Physics Vol. 36 nr. 1. 2010 Springer). However at least a neuronal basis for hyper-synchronized
gamma (γ)-activity which can be transmitted have been shown by these observations.
Summary A graded response to the frontal
electrical stimulation position during ECT-sessions can be recorded
in surface EEG at a distant parietal site. The responses trigger a
hyper-synchronized gamma (γ)-activity in a majority of the sessions.
This observation might offer a new and improved monitoring technique
for ECT and raises questions of this hyper-synchronized gamma (γ)-activity
plays a role for the treatment effect. Furthermore, it demonstrates
a neuronal basis for transmission of hyper-synchronized gamma (γ)-activity
in cortex. |